FDA APPROVAL

FDA Approves Neurovalens’ Modius Sleep Headset for Insomnia
The FDA has granted clearance to Neurovalens’ Modius Sleep device, offering a novel, drug-free solution for chronic insomnia in the ...

Phathom Triumphs with Dual FDA Nods for GERD Therapy Voquezna
Phathom Pharmaceuticals has achieved rapid success with its innovative heartburn medication Voquezna (vonoprazan), securing two FDA approvals in just three ...
Pfizer’s Penbraya: First vaccine to prevent all five types of meningitis gets FDA approval
Pfizer has secured a significant regulatory victory in the race to introduce the first five-pronged meningitis vaccine in the United ...

Pfizer gets FDA nod for Lorbrena-Vizimpro combo to treat advanced lung cancer
Pfizer is making strategic moves to reinforce its presence in the oncology arena, with the FDA granting approval for its ...

FDA Approves Biogen’s Biosimilar, Challenging Roche’s Actemra Dominance in the Market
Biogen has achieved a remarkable milestone while contemplating the future of its biosimilars division. The company proudly announced that its ...

Invitae gets FDA approval for blood test that detects hereditary cancer genes
Invitae has achieved a significant milestone with the FDA granting de novo clearance for its groundbreaking blood test. This test ...

Viatris and Ocuphire’s Ryzumvi gets FDA approval for reversing eye dilation
Viatris has achieved a significant milestone in its pursuit of reaching $1 billion in eye care sales by 2028. The ...

Jardiance gets FDA green light for chronic kidney disease, challenging AstraZeneca’s rival drug
Shortly after securing European approval for the treatment of chronic kidney disease (CKD), Eli Lilly and Boehringer Ingelheim have achieved ...
FDA Approves Temodar (Temozolomide) Label Updates for Anaplastic Astrocytoma Treatment
In a significant development, the FDA has granted approval for temozolomide (marketed as Temodar) to be used as adjuvant therapy ...

FDA Approves Ojjaara (Momelotinib) for Treating High-Risk Myelofibrosis with Anemia a new competitor of Jakafi
The FDA has granted approval for momelotinib, marketed as Ojjaara, as a groundbreaking treatment option for adult patients grappling with ...

FDA approves Beacon Biosignals’ headset that uses AI to monitor and improve sleep quality
Beacon Biosignals has achieved an impressive milestone shortly after acquiring EEG headset manufacturer Dreem. The company has received FDA clearance ...

Mallinckrodt Launches New FDA-Approved ADHD Drug Lisdexamfetamine
Mallinckrodt, a global specialty pharmaceutical company, has made waves with an announcement that’s set to reshape the Attention-Deficit/Hyperactivity Disorder (ADHD) ...

FDA Approves Novartis’ Tyruko: Revolutionizing MS Treatment Landscape Amidst Legal Drama
Stepping onto the stage with a flourish, Novartis has secured a resounding victory against Biogen in the realm of pharmaceutical ...
CAR T-Cell Therapy in Hematological Malignancies: A Breakthrough or a Bust?
Hematological malignancies are cancers that affect the blood, bone marrow, and lymph nodes. They include leukemia, lymphoma, and multiple myeloma. ...
FDA Approves Ingrezza (valbenazine) Capsules to Treat Chorea in Huntington’s Disease
Neurocrine Biosciences has achieved a significant milestone with the announcement of US Food and Drug Administration (FDA) approval for Ingrezza ...

FDA Grants Landmark Approval to Veopoz for the Treatment of Pediatric and Adult CHAPLE Disease
On August 18, 2023, Regeneron Pharmaceuticals announced a remarkable milestone. The US Food and Drug Administration (FDA) has granted approval ...

Forxiga Receives Green Light in China to Mitigate Cardiovascular Mortality and Hospitalization in Adults Battling Symptomatic Chronic Heart Failure
China’s National Medical Products Administration (NMPA) has granted approval to Forxiga (dapagliflozin) as a groundbreaking solution in the fight against ...

FDA Grants Approval to Ipsen’s Sohonos Capsules, the First-Ever Treatment for Individuals with Fibrodysplasia Ossificans Progressiva
In 2019, Ipsen made a significant investment of $1 billion to acquire Clementia Pharmaceuticals and its rare disease drug, palovarotene. ...

Delcath Triumphs After Years: FDA Grants Approval to Hepzato Kit for Treating Metastatic Uveal Melanoma
After enduring nearly a decade of setbacks, Delcath has finally achieved a regulatory milestone in the United States. Delaware-based Delcath ...

Revance into Therapeutic Realm with FDA Approval for Cervical Dystonia Treatment, Daxxify
Revance, a prominent player in the aesthetics market, has now made a strategic move into the promising therapeutic arena. Following ...

Johnson & Johnson Breaks Into Prostate Cancer Market with Akeega
After the FDA’s green light for AstraZeneca/Merck and Pfizer’s PARP inhibitors, Johnson & Johnson (J&J) has now secured its own ...

Amylyx Pharmaceuticals Shines with ALS Drug’s Relyvrio’s Strong Performance and Promising Path Forward
Amylyx Pharmaceuticals continues to impress the financial world with its amyotrophic lateral sclerosis (ALS) drug, Relyvrio. The medication, known by ...
Regeneron’s High-Dose Eylea Demonstrates Persistence as FDA Decision Looms
While awaiting an FDA decision regarding a higher-dose version of Eylea (aflibercept), Regeneron has consistently emphasized the durability of their ...

Johnson & Johnson Takes Lead in Bispecific Antibody Approval for Multiple Myeloma, Leaving Pfizer in the Dust
The FDA has granted accelerated approval to talquetamab-tgvs, branded as Talvey, for treating relapsed or refractory multiple myeloma in adults ...
FDA Approves Izervay for Geographic Atrophy
Source – Astellas Pharma Astellas Pharma has received FDA approval for Izervay (avacincaptad pegol intravitreal solution) for the treatment of geographic ...

FDA Grants Approval to Epcoritamab– Redefining the Future of DLBCL
In the world of cancer treatment, groundbreaking advancements are constantly being made, offering new hope for patients and their loved ...

FDA sets the stage for adcomm on Sanofi, AZ’s RSV prospect later this week
GSK and Pfizer have plans to introduce their adult respiratory syncytial virus (RSV) vaccines later this year, while Sanofi and ...
Pfizer’s RSV Vaccine ABRYSVO: A Breakthrough for the Industry and RSV Prevention
Pfizer’s RSV Vaccine: In a groundbreaking development, Pfizer’s FDA approval for its RSV vaccine, ABRYSVO, has not only positioned the ...
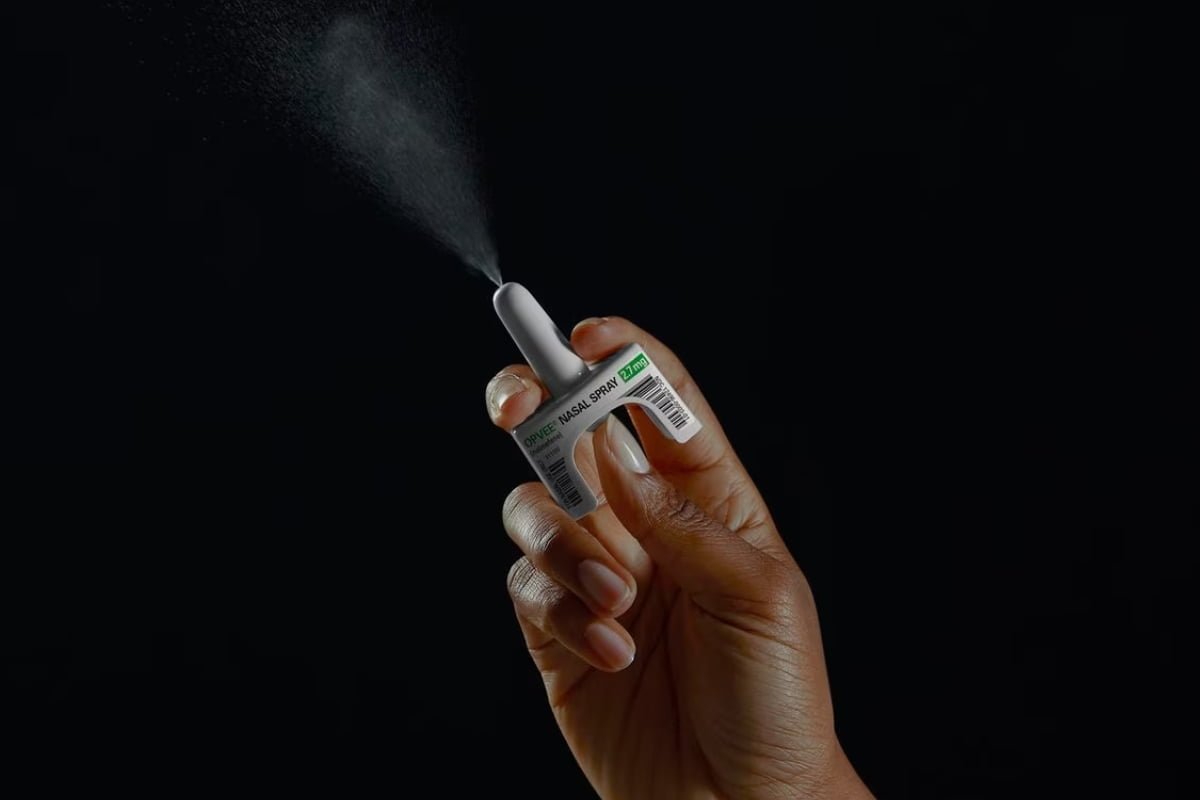
Indivior Receives FDA Approval for OPVEE (nalmefene)
Indivior’s OPVEE® (nalmefene) nasal spray has received FDA approval for the emergency treatment of opioid overdose in adults and pediatric ...