Daiichi Sankyo

Daiichi and AstraZeneca expand ADC pipelines after Enhertu triumph
At the European Society for Medical Oncology (ESMO) Congress, Daiichi Sankyo made a significant impact by striking a $4 billion ...

Dato-DXd Shows Efficacy in Lung Cancer, But Safety Issues Remain (ESMO 2023)
ESMO 2023: LBA12 During the Presidential Symposium at the ESMO Congress 2023 in Madrid, a study revealed that datopotamab deruxtecan, ...

Merck and Daiichi join forces to develop novel cancer drugs in $22B deal
In a strategic move, Merck & Co. has solidified its position in the thriving field of antibody-drug conjugates (ADCs) through ...

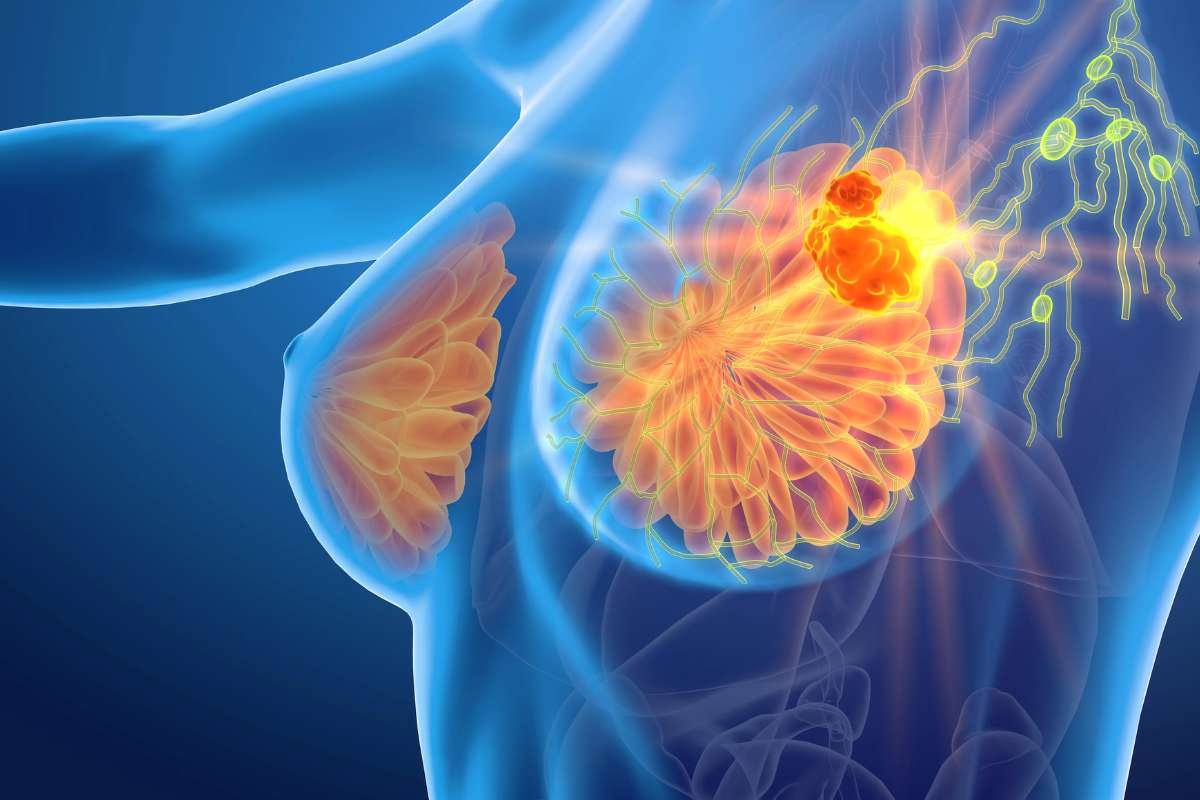
NICE rejects Enhertu for HER2-low breast cancer over cost concerns
The remarkable efficacy data that initially earned AstraZeneca and Daiichi Sankyo’s Enhertu a standing ovation at the ASCO meeting last ...

AstraZeneca and Daiichi Reach Key Milestone in Breast Cancer ADC Trial, Paving Way to Challenge Gilead
AstraZeneca and Daiichi Sankyo are making a strategic move to challenge Gilead Sciences’ position in the breast cancer arena. They ...
Enhertu, a Breakthrough Therapy for HER2 Mutant NSCLC, Gets EU Nod
Enhertu (trastuzumab deruxtecan) has received a positive recommendation for approval in the European Union (EU) as a standalone treatment for ...
Quizartinib Gets Green Light in EU for Treating Acute Myeloid Leukemia
Daiichi Sankyo has received a positive recommendation for the approval of quizartinib in the European Union (EU). This recommendation is ...

New drug Enhertu shows promise for HER2-mutant lung cancer patients (IASLC WCLC 2023 Conference)
The International Association for the Study of Lung Cancer (IASLC) presented pivotal results from the DESTINY-Lung02 Phase II trial at ...

FDA grants breakthrough status to T-DXd for two HER2-positive cancers
Daiichi Sankyo and AstraZeneca have garnered FDA Breakthrough Therapy Designations (BTDs) for their Enhertu (T-DXd) therapy to treat two distinct ...

Japanese Government Backs Arcalis with $115 Million Grants for mRNA Vaccine Production
Following Daiichi Sankyo’s recent accomplishment of securing Japan’s first mRNA vaccine approval, a potential competitor has emerged backed by significant ...

Daiichi Sankyo to Dominate Antibody-Drug Conjugates Market by 2029
Daiichi has successfully established ADCs (Antibody-Drug Conjugates) as a significant therapeutic approach, overcoming previous years of underwhelming results. Even though ...

Enhertu increases survival in solid tumors with HER2+
AstraZeneca and Daiichi Sankyo have received a boost in their efforts to gain approval for Enhertu as a therapy for ...

Kite Successfully Transfers Marketing Authorization for Yescarta CAR T-cell Therapy in Japan
Source – Gilead Sciences According to Kite Pharma on June 22, 2023, the marketing authorization for Yescarta, a chimeric antigen ...